SurgiCount
SurgiCount
End retained surgical sponges
one count at a time.
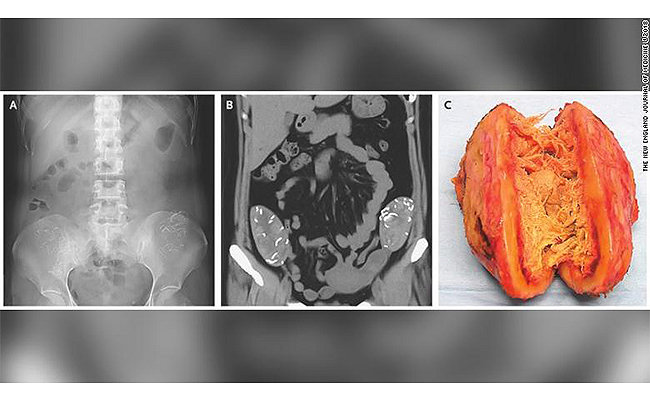
We’re on a mission to make healthcare better. This includes helping you in your fight against retained surgical sponges, the dangerous culprit most often left behind in cases of retained surgical items (RSI).1, 2, 7, 20, 21 Using the SurgiCount Safety-Sponge System as an adjunct process, our customers are avoiding the risk of the #1 surgical “never event,”3 and we’re confident you can too. That’s why it’s backed by an industry-unique indemnity program.

Straight from the source
Listen to what our customers have to say.
9100-003-611 Rev None
Stryker Corporation or its divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: SAFE-T Lap, Safety-Sponge, Stryker, SurgiCount, SurgiCount360, and SurgiCounter. All other trademarks are trademarks of their respective owners or holders.
9100-003-611 Rev None